LDT and IHD Diagnostics Regulation: What Changed and What to Do Next
- Mar 6
- 6 min read
How U.S. and EU Decisions Are Reshaping Your Market Strategy
by David Petrich and Pieter Bogaert
If you're developing in vitro diagnostics (IVDs) for global markets, you just experienced one of the most volatile regulatory years in a decade. In the U.S., a federal court overturned the FDA's proposed oversight of Lab-Developed Tests (LDTs). In Europe, regulators moved in the opposite direction, tightening requirements for In-House Devices (IHDs).
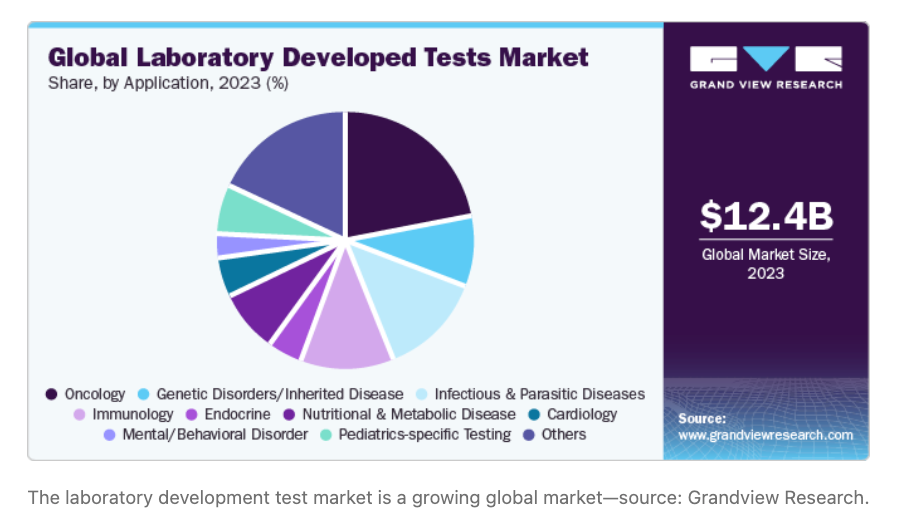
For startups, manufacturers, and clinical labs, this divergence creates both risk and opportunity. The U.S. and EU still account for half of global clinical trials, despite the rapid rise of trials in China and other Asian nations. How these regulatory frameworks evolve directly impacts time-to-market, trial feasibility, and development costs for diagnostic companies and clinical laboratories worldwide. The overall market for these types of tests exceeds $12B and poised to grow significantly over the next decade, supported by applications in oncology, genetic disorders, infectious disease, and other conditions.
Here's what changed, and what you need to do about it.
Understanding the Products at the Center of Changes
Laboratory Developed Tests (LDTs) in the U.S. and In-House Devices (IHDs) in Europe are diagnostic tests manufactured and used within a single laboratory or health institution. They encompass several distinct categories:
• True homebrew tests: designed, manufactured, validated, and used exclusively within a single laboratory.
• Modified commercial IVDs: commercially available tests that a lab adapts for a specific clinical need.
• Research-grade assays repurposed for clinical use: used to analyze biomarkers or analytes in drug development and preclinical studies.
All three categories have historically operated under light-touch regulatory oversight. That era is now winding down at different speeds and in different directions depending on the jurisdiction.
The Regulatory Roadmap Divergence
The U.S. Reversal
Since 2024, the FDA communicated a course toward stricter oversight. The agency had proposed that laboratories comply with Quality Management Systems (QMS), device and establishment registration, complaint tracking, and pre-market submissions like commercial IVD manufacturers.
Then, in March 2025, a federal judge halted the rule, determining the FDA had exceeded its statutory authority. Next, the FDA formally rescinded the regulation in August 2025, thereby vacating the ‘LDT Rule’, which would regulate LDTs as medical devices[DP1] .
The result? Regulatory uncertainty. LDTs remain largely unsupervised, but future FDA action is possible. Litigation is still pending. Organizations that built compliance programs in anticipation of the new rule now face a strategic recalibration.
The EU's Opposite Path – with a Key Wild Card
European Union (EU) In Vitro Diagnostic Regulation (IVDR) is tightening step-by-step through 2029. Laboratories that make and use IHDs must now comply with comprehensive QMS requirements, risk assessments, performance evaluations, and follow-up of IHD performance under Article 5.5 – even though they're technically exempt from some rules for commercial IVD manufacturers.
One important development: in December 2025, the European Commission published a proposal to remove the restriction that IHDs can only be used when no equivalent CE-marked device exists. If adopted, this change would significantly broaden the scenarios in which IHDs are permissible — fundamentally altering the competitive landscape for both labs and commercial manufacturers. The proposal remains subject to final implementation, so uncertainty persists, but it signals a meaningful shift in regulatory intent[DP2] [PB3] .
The Hidden Impact: Clinical Trials and Innovation
Neither the U.S. nor the EU regulatory path is academic. Both are affecting the balance between safe and effective diagnostic tests and products, and real innovation used to improve patient healthcare.
In the EU, IVDR's strict requirement that diagnostic tests be either CE-marked or fully compliant IHDs has created unexpected friction in pharmaceutical development. Clinical performance evaluation studies or clinical trials relying on diagnostic tests from U.S. labs (operating as LDTs) now face regulatory mismatches. This stifled pharma innovation enough that the European Commission launched pilot programs to streamline the use of such tests in clinical trials.
The lesson: Regulatory categorization directly affects time-to-market, clinical trial feasibility, and product development costs. Getting ahead of these constraints — rather than discovering them during trial setup — is increasingly a competitive differentiator.

Three Strategic Paths Forward
Your diagnostic product or test likely follows one of three trajectories:
Path 1: Mass-Market Commercialization
Examples like Foundation One's NGS panel, Mayo Clinic's MASS-FIX, and LabCorp's pTau-217 assay started as institutional LDTs and evolved into commercial IVD products serving millions of patients. If this is your goal, regulatory rigor from day one becomes a competitive advantage.
Path 2: Narrow Clinical Use Cases
Some tests – particularly in precision medicine and rare disease – are intentionally designed for individual lab settings without plans for broad commercialization. These still require robust QMS frameworks, but the scale is different.
Path 3: Short-Term Applications
Clinical Trial Assays (CTAs), Companion Diagnostics (CDx), and short-term diagnostic solutions have defined lifespans. Tighter LDT regulation in the U.S. and stricter IHD oversight in Europe will compress the timeline and raise costs for these products.
Building for Optionality: The Foundational Framework
Facing regulatory uncertainty, the smartest move is to build with optionality. Whether your diagnostic test becomes a commercial IVD or remains an LDT or IHD, these foundational practices protect your investment and keep your options open.
On the Design Side
Define your intended use from day one: analyte, specimen type, target population, clinical setting, and platform. This clarity – which many labs skip – becomes invaluable if you later seek FDA approval in the U.S. or CE marking in the EU.
Create a controlled assay configuration: document your protocol, instruments, reagent lots, software, and algorithms. Manage any changes through formal design control (e.g., requirements, verification, validation, risk assessment). Treat your LDT/IHD as if you're building a Design History File – because you might be.
Why? Design control and risk management are integrated, life cycle processes that are expensive and difficult to retrofit or redo. Implementing both processes early prevents costly reconstruction later. Design control produces safer and more characterized tests and products. This is good business practice, independent of the regulatory path you ultimately choose.
On the Data Side
Generate device-grade validation, not minimum CLIA evidence. This means comprehensive analytical validation: detection limits, linearity, precision, accuracy against a comparator, specificity, interference testing, stability, and full traceability of raw data.
Use harmonized consensus standards (like CLSI) in your study designs. This ensures your data will be accepted by the FDA, EU Notified Bodies, and other regulators globally. If you do not use or modify harmonized consensus standards, you must document your justification.
For clinical validation, pre-specify your protocol and statistical analysis. Define your target population, select appropriate predicate devices or comparators (FDA-cleared tests or accepted clinical standards), and organize outputs in submission-ready reports.
Documentation and Quality Systems
Implement a digital, modular QMS early in the test or product development process. Include design control and risk management as integrated lifecycle processes. Good documentation practices from day one prevent the need to scramble to recreate data and decisions later. Many digital QMS are commercially available, which include AI and other automated features to improve the efficiency and effectiveness of a diagnostic test or product development program.

EU-Specific Considerations
If the EU is part of your market, add these requirements to your framework:
Your design must explicitly trace to IVDR's General Safety and Performance Requirements (Annex I). Map every design decision, risk mitigation, and residual risk to these requirements.
Clinical evidence under IVDR requires a formal performance evaluation documenting scientific validity, analytical performance, and clinical performance. Unlike FDA 510(k) submissions, you cannot rely on substantial equivalence to a predicate device – you must generate absolute evidence of clinical performance (though method comparison is acceptable when ground truth cannot be established independently).
Post-market surveillance isn't optional. Your conformity assessment must include a plan for continuous performance follow-up and surveillance.
The Bottom Line for Your Organization
The regulatory environment is uncertain and diverging. The U.S. offers short-term flexibility but long-term unpredictability. The EU is moving toward formalization with a clear — if demanding — pathway. Either way, the global trend favors rigor over shortcuts.
The business case for building to a device-grade quality standard is strong. Investing in design control, data integrity, documentation, and a digital QMS does four things simultaneously:
• Reduces regulatory risk and costly rework downstream.
• Compresses time-to-market if commercialization becomes the goal.
• Preserves optionality as regulations continue to evolve.
• Positions products to compete globally, not just in a single market.
The diagnostic tests that will thrive in the next decade won't be those that cut corners under outdated exemptions. They'll be those built on a foundation of design excellence and data integrity from day one.
David Petrich is co-founder and VP, Quality and Regulatory, of Landrich Group, based in the San Francisco Bay Area. He specializes in regulatory strategy for in vitro diagnostics and laboratory-developed tests.
Pieter Bogaert, based in Belgium, is Head of Quality and Regulatory of hema.to GmbH, an AI cytometry company focused on advanced precision immune medicine. He is also co-chair of RAPS (Regulatory Affairs Professional Society) Belgium.
What's your team's biggest challenge in navigating these regulatory shifts? We’d like to hear how LDT/IHD regulations are affecting your product roadmap in the comments below.




